Generic Drugs: What They Are, Why They Work, and When They Don’t
When you pick up a prescription, you might see generic drugs, lower-cost versions of brand-name medications that contain the same active ingredient. Also known as generic medications, they’re required by law to work the same way as the original—same dose, same route, same effect. But here’s the catch: they don’t have to be identical in every way. The FDA says they’re bioequivalent, meaning they deliver the same amount of active drug into your bloodstream within the same timeframe. That’s true for most people. But for some, even tiny differences in fillers, coatings, or how the pill breaks down can make a real difference.
That’s why inactive ingredients, the non-medical parts of a pill like dyes, binders, or fillers. Also known as excipients, they’re not just harmless additives. For people with sensitivities, allergies, or conditions like celiac disease, these can trigger reactions—even if the active drug is perfectly matched. And when it comes to drugs with a narrow therapeutic index, medications where even small changes in blood levels can cause serious harm. Also known as NTI drugs, they include warfarin, levothyroxine, and some seizure meds, switching between generic brands can sometimes cause your levels to swing too high or too low. One study found that patients on levothyroxine who switched generics had noticeable changes in their TSH levels, even when the active ingredient was the same.
It’s not about quality—it’s about consistency. Brand-name drugs are made with the same formula, same factory, same batch process every time. Generics? They’re made by different companies, sometimes in different countries, using different processes. That’s why your doctor might ask you to stick with one brand of generic, or even stick with the original if you’re on a sensitive medication. It’s not that generics are bad. They’re often safe, effective, and save you hundreds a year. But they’re not magic clones. Your body might notice the difference, especially if you’re taking multiple meds, have a chronic condition, or are sensitive to changes in how things feel.
What you’ll find below are real stories and science-backed insights from people who’ve been there: why some switched from brand to generic and felt worse, how fiber can mess with absorption, what hidden animal ingredients are in pills, and why your thyroid med might need special handling. These aren’t theoretical debates—they’re lived experiences, backed by data, and focused on what actually matters: keeping you healthy, safe, and in control of your treatment.
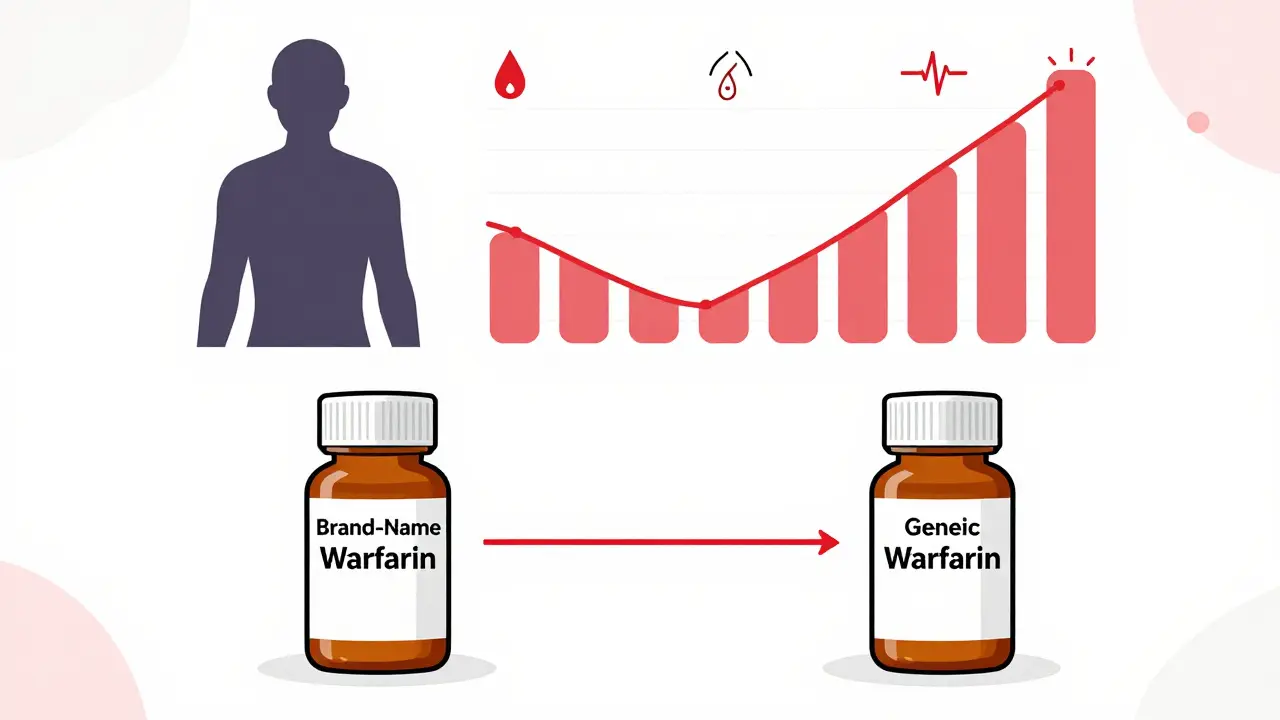
International Perspectives on NTI Generics: Regulatory Approaches Compared
NTI generics require strict regulatory oversight due to their narrow safety margin. This article compares how the U.S., EU, Canada, and Japan regulate these high-risk drugs, and why global consistency still lags behind patient needs.
Read More
History of Generic Drugs in the United States
Generic drugs now make up over 90% of U.S. prescriptions but cost only 23% of total drug spending. Their history traces back to 1820, with key laws like Hatch-Waxman and FDA reforms shaping today's affordable medication system.
Read More
Authorized Generics: A Smarter Switch When Leaving Brand-Name Drugs
Authorized generics offer the exact same medication as brand-name drugs at lower prices, with no changes to ingredients or effectiveness. Learn how they help patients avoid side effects and save money.
Read More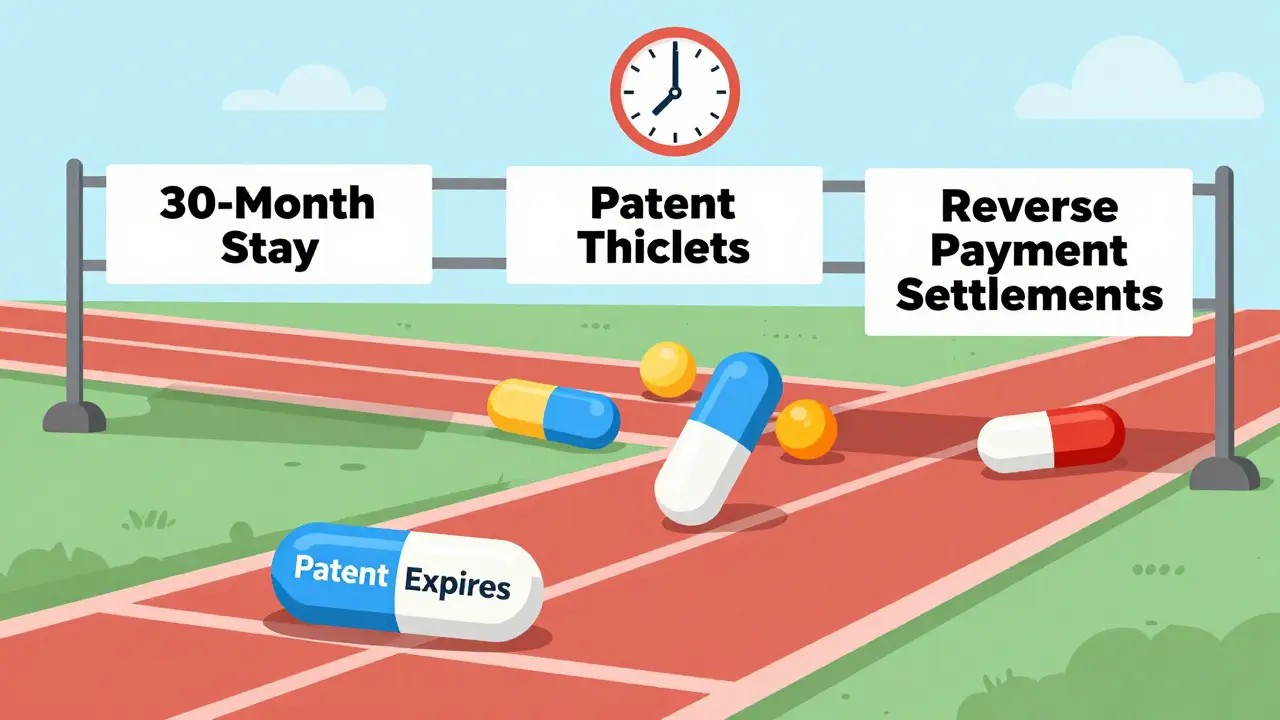
Generic Drug Availability: From Patent Expiration to Market Launch
Generic drugs don't launch right after patents expire. Legal delays, patent thickets, and corporate tactics create years-long gaps between expiration and availability - costing patients billions. Here's how it really works.
Read More
How Other Countries See Generics: Global Provider Perspectives
Generics are viewed differently around the world-from essential tools in Europe to lifelines in Asia and risky bets in the U.S. This article explores how providers in key countries see generic drugs and why their perspectives shape global access to medicine.
Read More
How to Effectively Discuss Generic Medications with Patients
Learn how to clearly explain generic medications to patients, address common concerns, and improve adherence using proven communication strategies backed by clinical evidence and real-world data.
Read More
Coverage of Generics vs Brands: Key Policy Differences in Insurance Formularies
Insurance policies treat generic and brand-name drugs very differently, with generics costing up to 85% less. Learn how formularies, substitution rules, prior authorization, and state laws shape what you pay-and what you actually get.
Read More
Addressing Patient Concerns About Generic Medications: Common Questions Answered
Generic medications are just as safe and effective as brand-name drugs, but many patients still have questions. This guide answers the most common concerns about generics-look, cost, side effects, and when to stick with brand-name.
Read More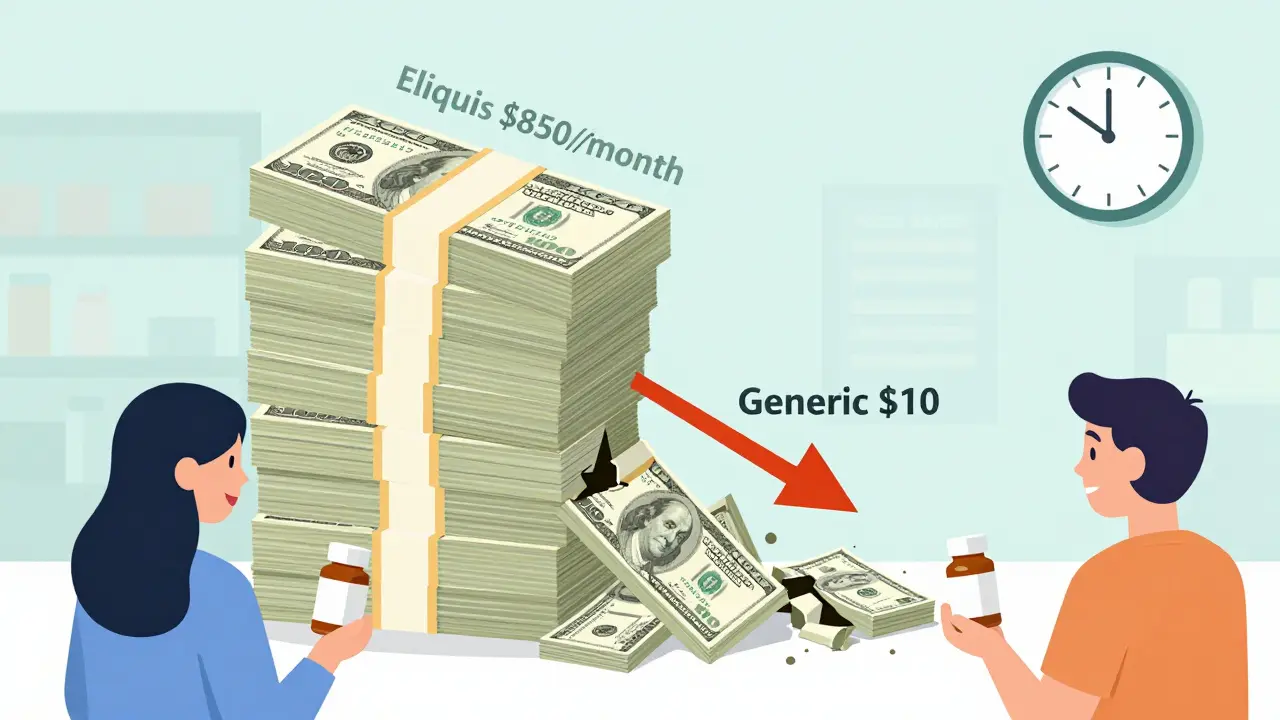
Economic Impact of Patent Expiration: When Drug Prices Drop
When pharmaceutical patents expire, drug prices drop dramatically-often by 80% or more. This article explains how generic competition drives down costs, why some drugs stay expensive despite patent expiry, and what patients can do to save money.
Read More
Provider Education on Generics: How Clinicians Can Improve Patient Outcomes and Cut Costs
Generic drugs make up 90% of prescriptions but many clinicians still doubt their effectiveness. Learn how provider education closes knowledge gaps, improves patient adherence, and saves billions in healthcare costs.
Read More